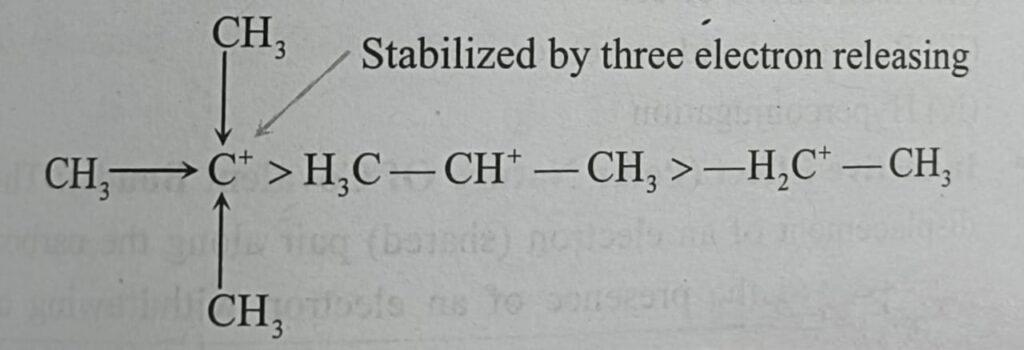The study of carbon-containing compounds and their reaction.
Topic :-
- Basic Concepts of Organic Chemistry and Stereochemistry
- Organic Reaction Mechanism and Synthetic Applications
- Qualitative Organic Analysis
- Natural Products Chemistry
- Aromatic and Heterocyclic Chemistry
Basic Concepts of Organic Chemistry and Stereochemistry
- Basic Concepts in Organic Chemistry
- Resonance and Mesmeric Effect
- Hyperconjugation
- Types of Organic Reactions
- Nomenclature
- Isomerism
- Hydrocarbons
- Alkane or Paraffins
- Alkenes or Olefins
- Alkynes
Basic Concepts in Organic Chemistry
Reaction Intermediate :–
Most of organic reactions occur through the involvement of certain chemical species. These are generally short lived (106 seconds to a few seconds ) and highly reactive and hence cannot be isolated. These short lived highly reactive chemical species. Through which the majority of the organic reactions occur are called reactive intermediates these intermediates are detected by spectroscopic methods or trapped chemically or their presence is confirmed by indirect evidence. On the other hand, synthetic intermediate are stable products which are prepared isolated and purified and subsequently used as starting materials in a synthetic sequence.
Carbocations (Earlier Called as Carbonium Ions) :-
Carbocations are the key intermediates in several reactions and particularly in nucleophilic substitution reactions and electrophilic addition reaction.
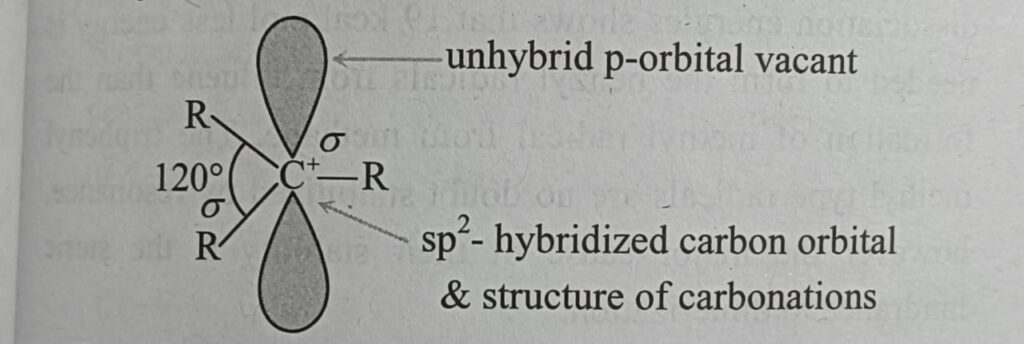
Structure :-
Generally in the carbocations the positively charged carbon atom is bonded to three others atoms and has no nonbonding electrons. It is sp2 hybridized with a planer structure and bond angles are of about 120o . There is a vacant unhybridized p orbital which (e.g in the case of CH3 ) lies perpendicular to the plane of C – H bonds.
PRABHAT PAPERBACKS
Stability :-
There is an increase in carbocation stability with additional alky substitution . Thus one finds that addition of HX to three typical olefins decreases in the order (CH3)2C = CH2 >CH3 – CH = CH2 >CH2 = CH2 This is due to the relative stabilities of the carbocations formed in the rate determining step which in turn follows from the fact that the stability is increased by the electron releasing methyl group (+I), three such groups being more effective than two, and two more effective than one.